Iron(III) chloride
Videos
Page
Iron(III) chloride describes the inorganic compounds with the formula FeCl3(H2O)x. Also called ferric chloride, these compounds are some of the most important and commonplace compounds of iron. They are available both in anhydrous and in hydrated forms which are both hygroscopic. They feature iron in its +3 oxidation state. The anhydrous derivative is a Lewis acid, while all forms are mild oxidizing agents. It is used as a water cleaner and as an etchant for metals.

Image: Iron(III) chloride anhydrate

Image: Хлорид железа
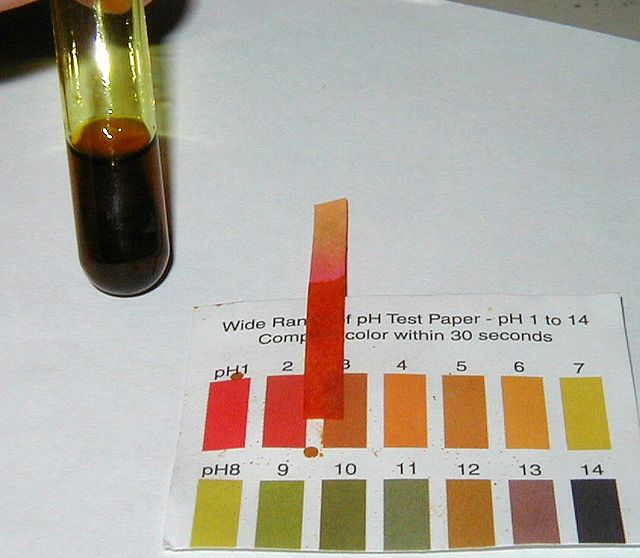
A brown, acidic solution of iron(III) chloride.
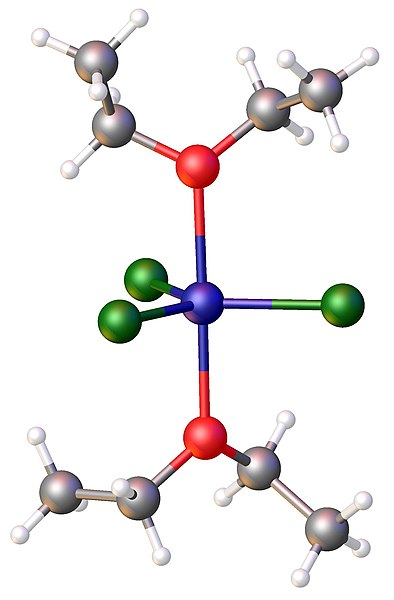
Structure of FeCl3(diethylether)2. Color code: Cl=green,Fe = blue, O = red.
Iron
Videos
Page
Iron is a chemical element; it has symbol Fe and atomic number 26. It is a metal that belongs to the first transition series and group 8 of the periodic table. It is, by mass, the most common element on Earth, forming much of Earth's outer and inner core. It is the fourth most common element in the Earth's crust, being mainly deposited by meteorites in its metallic state.

Iron

A polished and chemically etched piece of an iron meteorite, believed to be similar in composition to the Earth's metallic core, showing individual crystals of the iron-nickel alloy (Widmanstatten pattern)

Ochre path in Roussillon

Banded iron formation in McKinley Park, Minnesota