Sulfonamide (medicine)
Videos
Page
Sulfonamide is a functional group that is the basis of several groups of drugs, which are called sulphonamides, sulfa drugs or sulpha drugs. The original antibacterial sulfonamides are synthetic (nonantibiotic) antimicrobial agents that contain the sulfonamide group. Some sulfonamides are also devoid of antibacterial activity, e.g., the anticonvulsant sultiame. The sulfonylureas and thiazide diuretics are newer drug groups based upon the antibacterial sulfonamides.

Person with Stevens–Johnson syndrome
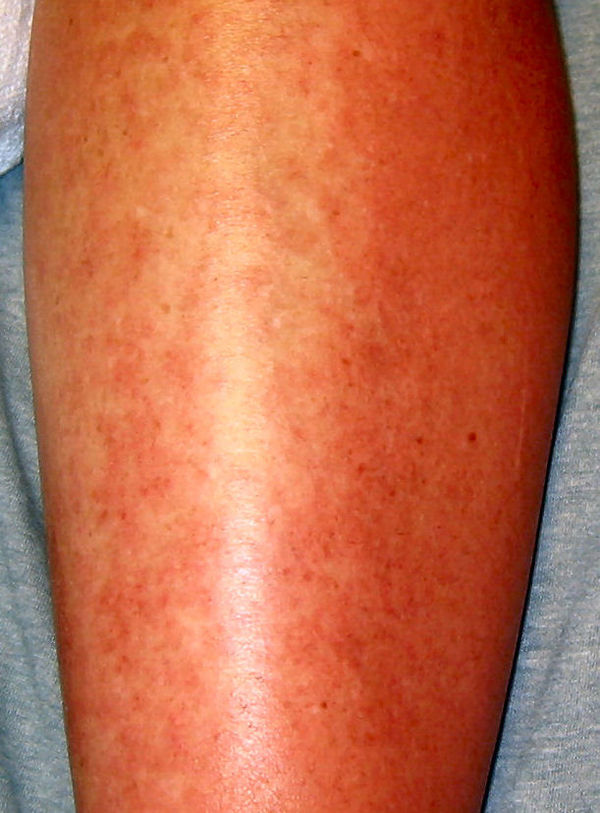
Allergic urticaria on the skin induced by an antibiotic
Bayer
Videos
Page
Bayer AG is a German multinational pharmaceutical and biotechnology company and is one of the largest pharmaceutical companies and biomedical companies in the world. Headquartered in Leverkusen, Bayer's areas of business include: pharmaceuticals, consumer healthcare products, agricultural chemicals, seeds and biotechnology products. The company is a component of the EURO STOXX 50 stock market index.

Headquarters in Leverkusen

Share of Farbenfabriken vorm. Friedr. Bayer & Comp in Elberfeld, issued 1 May 1908

Bayer cross, Leverkusen

Bottle of Bayer aspirin, 1899