Carbonatation
Videos
Page
Carbonatation is a chemical reaction in which calcium hydroxide reacts with carbon dioxide and forms insoluble calcium carbonate:

Carbonatation induced rebar corrosion
Calcium hydroxide
Videos
Page
Calcium hydroxide (traditionally called slaked lime) is an inorganic compound with the chemical formula Ca(OH)2. It is a colorless crystal or white powder and is produced when quicklime (calcium oxide) is mixed with water. It has many names including hydrated lime, caustic lime, builders' lime, slaked lime, cal, and pickling lime. Calcium hydroxide is used in many applications, including food preparation, where it has been identified as E number E526. Limewater, also called milk of lime, is the common name for a saturated solution of calcium hydroxide.

Image: Calcium hydroxide
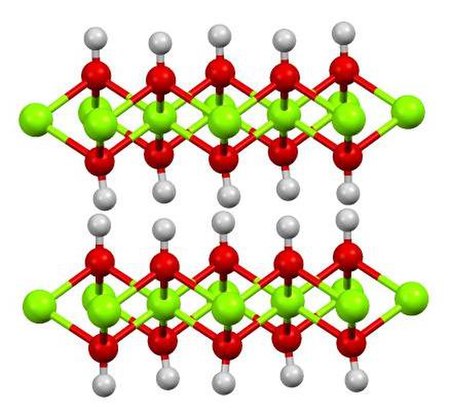
Image: Mg(OH)2Xray

SEM image of fractured hardened cement paste, showing plates of calcium hydroxide and needles of ettringite (micron scale)

Dry untreated maize (left), and treated maize (right) after boiling in water with calcium hydroxide (15 ml, or 1 tbsp, lime for 500 g of corn) for 15 minutes