Diamond
Videos
Page
Diamond is a solid form of the element carbon with its atoms arranged in a crystal structure called diamond cubic. Another solid form of carbon known as graphite is the chemically stable form of carbon at room temperature and pressure, but diamond is metastable and converts to it at a negligible rate under those conditions. Diamond has the highest hardness and thermal conductivity of any natural material, properties that are used in major industrial applications such as cutting and polishing tools. They are also the reason that diamond anvil cells can subject materials to pressures found deep in the Earth.

A naturally-cut diamond crystal
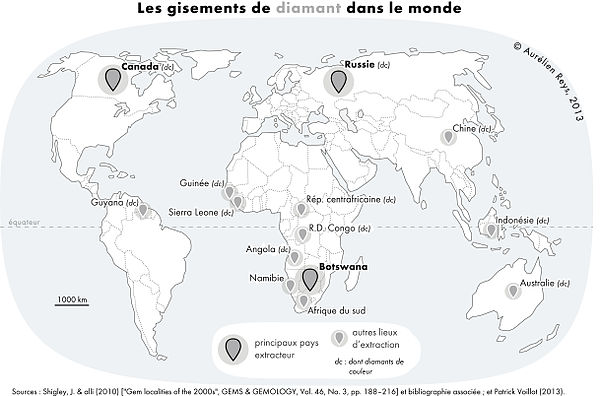
Main diamond producing countries

One face of an uncut octahedral diamond, showing trigons (of positive and negative relief) formed by natural chemical etching

The extreme hardness of diamond in certain orientations makes it useful in materials science, as in this pyramidal diamond embedded in the working surface of a Vickers hardness tester.
Allotropes of carbon
Videos
Page
Carbon is capable of forming many allotropes due to its valency. Well-known forms of carbon include diamond and graphite. In recent decades, many more allotropes have been discovered and researched, including ball shapes such as buckminsterfullerene and sheets such as graphene. Larger-scale structures of carbon include nanotubes, nanobuds and nanoribbons. Other unusual forms of carbon exist at very high temperatures or extreme pressures. Around 500 hypothetical 3‑periodic allotropes of carbon are known at the present time, according to the Samara Carbon Allotrope Database (SACADA).

Computer models of stable nanobud structures

A large sample of glassy carbon.
The K4 crystal

Diamond and graphite are two allotropes of carbon: pure forms of the same element that differ in structure.