Heavy water
Videos
Page
Heavy water is a form of water whose hydrogen atoms are all deuterium rather than the common hydrogen-1 isotope that makes up most of the hydrogen in normal water. The presence of the heavier isotope gives the water different nuclear properties, and the increase in mass gives it slightly different physical and chemical properties when compared to normal water.
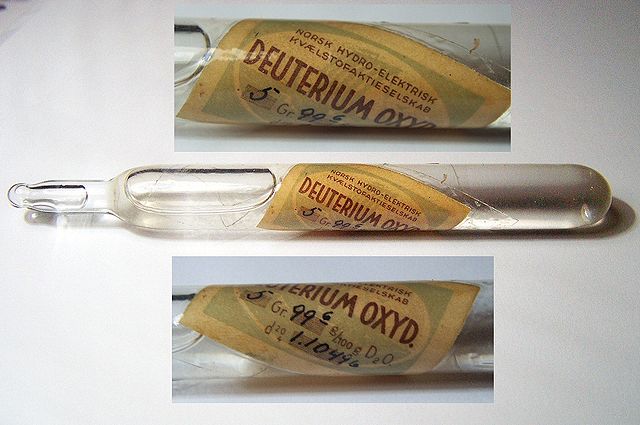
"Heavy water" made by Norsk Hydro
Deuterium
Videos
Page
Deuterium (hydrogen-2, symbol 2H or D, also known as heavy hydrogen) is one of two stable isotopes of hydrogen (the other is protium, or hydrogen-1). The deuterium nucleus, called a deuteron, contains one proton and one neutron, whereas the far more common protium has no neutrons in the nucleus. Deuterium has a natural abundance in Earth's oceans of about one atom of deuterium among every 6,420 atoms of hydrogen (see heavy water). Thus deuterium accounts for about 0.0156% by number (0.0312% by mass) of all hydrogen in the oceans: 4.85×1013 tonnes of deuterium – mainly in form of HOD (or 1HO2H or 1H2HO) and only rarely in form of D2O (or 2H2O) – in 1.4×1018 tonnes of water. The abundance of deuterium changes slightly from one kind of natural water to another (see Vienna Standard Mean Ocean Water).

Deuterium discharge tube

Ionized deuterium in a fusor reactor giving off its characteristic pinkish-red glow

Harold Urey, deuterium's discoverer

The "Sausage" device casing of the Ivy Mike H bomb, attached to instrumentation and cryogenic equipment. The 20-ft-tall bomb held a cryogenic Dewar flask with room for 160 kg of liquid deuterium.