History of the periodic table
Videos
Page
The periodic table is an arrangement of the chemical elements, structured by their atomic number, electron configuration and recurring chemical properties. In the basic form, elements are presented in order of increasing atomic number, in the reading sequence. Then, rows and columns are created by starting new rows and inserting blank cells, so that rows (periods) and columns (groups) show elements with recurring properties. For example, all elements in group (column) 18 are noble gases that are largely—though not completely—unreactive.

The American chemist Glenn T. Seaborg—after whom the element seaborgium is named—standing in front of a periodic table, May 19, 1950

Hennig Brand, as shown in The Alchemist Discovering Phosphorus
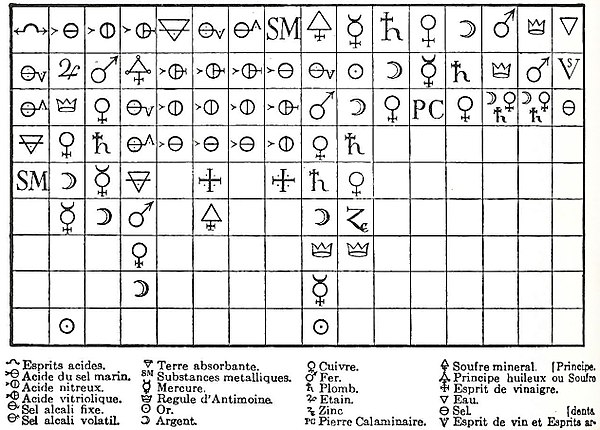
Geoffroy's 1718 Affinity Table: at the head of each column is a chemical species with which all the species below can combine. Some historians have defined this table as being the start of the chemical revolution.

Dalton (1806): listing the known elements by atomic weight
Atomic number
Videos
Page
The atomic number or nuclear charge number of a chemical element is the charge number of an atomic nucleus. For ordinary nuclei composed of protons and neutrons, this is equal to the proton number (np) or the number of protons found in the nucleus of every atom of that element. The atomic number can be used to uniquely identify ordinary chemical elements. In an ordinary uncharged atom, the atomic number is also equal to the number of electrons.

Russian chemist Dmitri Mendeleev, creator of the periodic table.

Niels Bohr, creator of the Bohr model.

Henry Moseley in his lab.