Supercritical fluid
Videos
Page
A supercritical fluid (SCF) is any substance at a temperature and pressure above its critical point, where distinct liquid and gas phases do not exist, but below the pressure required to compress it into a solid. It can effuse through porous solids like a gas, overcoming the mass transfer limitations that slow liquid transport through such materials. SCF are superior to gases in their ability to dissolve materials like liquids or solids. Also, near the critical point, small changes in pressure or temperature result in large changes in density, allowing many properties of a supercritical fluid to be "fine-tuned".

Figure 2. Carbon dioxide density-pressure phase diagram
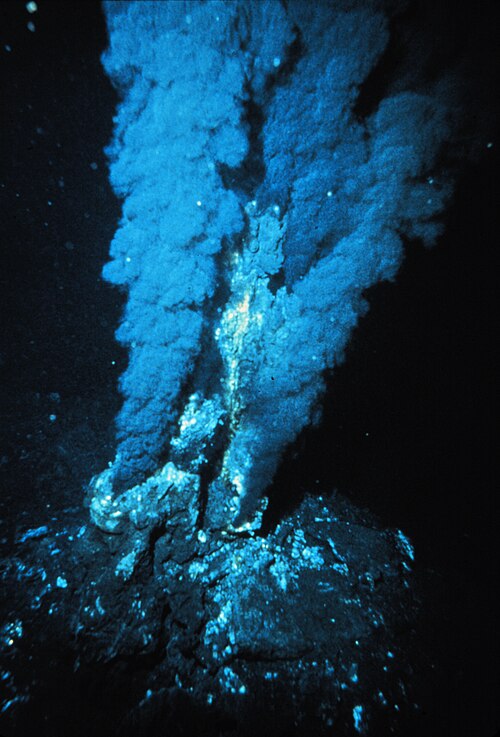
A black smoker, a type of hydrothermal vent
Critical point (thermodynamics)
Videos
Page
In thermodynamics, a critical point is the end point of a phase equilibrium curve. One example is the liquid–vapor critical point, the end point of the pressure–temperature curve that designates conditions under which a liquid and its vapor can coexist. At higher temperatures, the gas cannot be liquefied by pressure alone. At the critical point, defined by a critical temperature Tc and a critical pressure pc, phase boundaries vanish. Other examples include the liquid–liquid critical points in mixtures, and the ferromagnet–paramagnet transition in the absence of an external magnetic field.

Subcritical ethane, liquid and gas phase coexist. Critical point (32.17 °C, 48.72 bar), opalescence. Supercritical ethane, fluid.

Critical carbon dioxide exuding fog while cooling from supercritical to critical temperature.