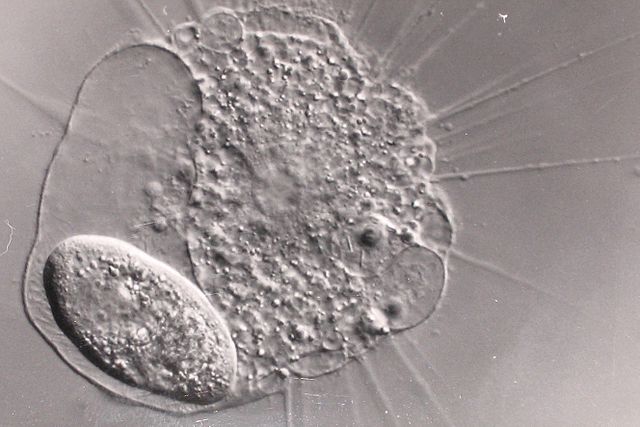
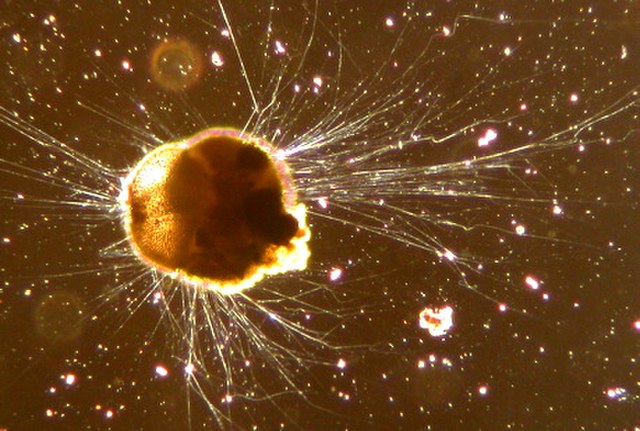
The actinophryids are an order of heliozoa, a polyphyletic array of stramenopiles, having a close relationship with pedinellids and Ciliophrys. They are common in fresh water and occasionally found in marine and soil habitats. Actinophryids are unicellular and roughly spherical in shape, with many axopodia that radiate outward from the cell body. Axopodia are a type of pseudopodia that are supported by hundreds of microtubules arranged in interlocking spirals and forming a needle-like internal structure or axoneme. Small granules, extrusomes, that lie under the membrane of the body and axopodia capture flagellates, ciliates and small metazoa that make contact with the arms.
Actinophryid
Cross-section of the double spiral microtubule structure in an axopod
Actinophrys undergoing multiple plasmotomy
Actinophrys and Paramecium
An amoeba, often called an amoeboid, is a type of cell or unicellular organism with the ability to alter its shape, primarily by extending and retracting pseudopods. Amoebae do not form a single taxonomic group; instead, they are found in every major lineage of eukaryotic organisms. Amoeboid cells occur not only among the protozoa, but also in fungi, algae, and animals.
Clockwise from top right: Amoeba proteus, Actinophrys sol, Acanthamoeba sp., Nuclearia thermophila., Euglypha acanthophora, neutrophil ingesting bacteria.
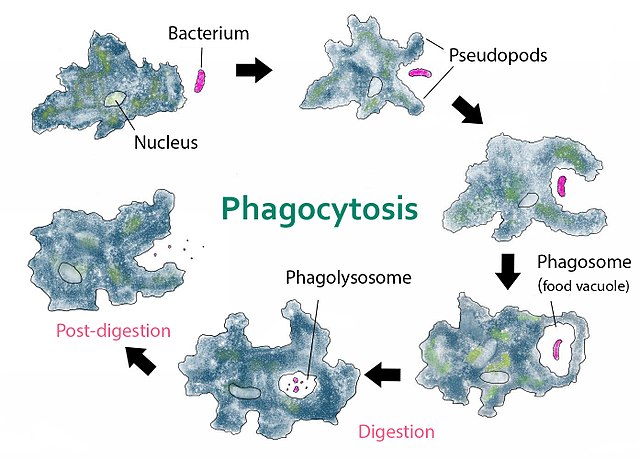
Amoeba phagocytosis of a bacterium
Foraminifera have reticulose (net-like) pseudopods, and many species are visible with the naked eye
Neutrophil (white blood cell) engulfing anthrax bacteria