The Antarctic ice sheet is a continental glacier covering 98% of the Antarctic continent, with an area of 14 million square kilometres and an average thickness of over 2 kilometres (1.2 mi). It is the largest of Earth's two current ice sheets, containing 26.5 million cubic kilometres of ice, which is equivalent to 61% of all fresh water on Earth. Its surface is nearly continuous, and the only ice-free areas on the continent are the dry valleys, nunataks of the Antarctic mountain ranges, and sparse coastal bedrock. However, it is often subdivided into East Antarctic ice sheet (EAIS), West Antarctic ice sheet (WAIS), and Antarctic Peninsula (AP), due to the large differences in topography, ice flow, and glacier mass balance between the three regions.
South facing visualization of the Antarctic ice sheet from the Pacific sector of the Southern Ocean (West Antarctic ice sheet, foreground; Antarctic Peninsula, to the left; East Antarctic ice sheet, background).
Antarctic Skin Temperature Trends between 1981 and 2007, based on thermal infrared observations made by a series of NOAA satellite sensors. Skin temperature trends do not necessarily reflect air temperature trends.
Parts of East Antarctica (marked in blue) are currently the only place on Earth to regularly experience negative greenhouse effect during certain months of the year. At greater warming levels, this effect is likely to disappear due to increasing concentrations of water vapor over Antarctica
Antarctic surface temperature trends, in °C/decade. Red represents areas where temperatures have increased the most since the 1950s.
In glaciology, an ice sheet, also known as a continental glacier, is a mass of glacial ice that covers surrounding terrain and is greater than 50,000 km2 (19,000 sq mi). The only current ice sheets are the Antarctic ice sheet and the Greenland ice sheet. Ice sheets are bigger than ice shelves or alpine glaciers. Masses of ice covering less than 50,000 km2 are termed an ice cap. An ice cap will typically feed a series of glaciers around its periphery.
One of Earth's two ice sheets: The Antarctic ice sheet covers about 98% of the Antarctic continent and is the largest single mass of ice on Earth. It has an average thickness of over 2 kilometers.
Glacial flow rate in the Antarctic ice sheet.
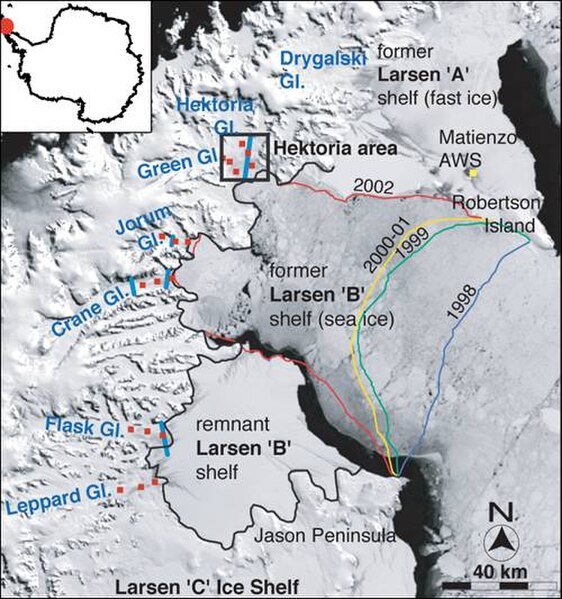
The collapse of the Larsen B ice shelf had profound effects on the velocities of its feeder glaciers.
Accelerated ice flows after the break-up of an ice shelf