Blood vessel
Videos
Page
Blood vessels are the components of the circulatory system that transport blood throughout the human body. These vessels transport blood cells, nutrients, and oxygen to the tissues of the body. They also take waste and carbon dioxide away from the tissues. Blood vessels are needed to sustain life, because all of the body's tissues rely on their functionality.
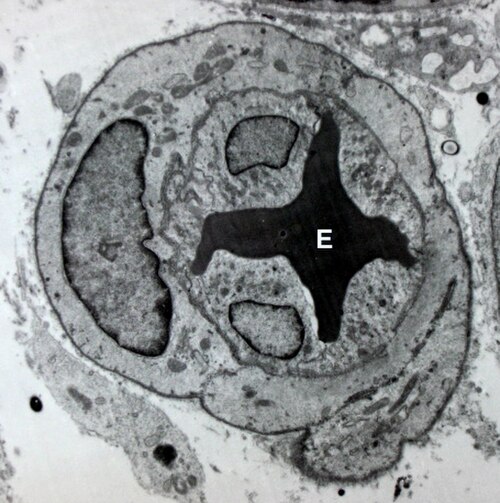
Transmission electron micrograph of a blood vessel displaying an erythrocyte (red blood cell, E) within its lumen, endothelial cells forming its tunica intima (inner layer), and pericytes forming its tunica adventitia (outer layer).

Constricted blood vessel.
Blood
Videos
Page
Blood is a body fluid in the circulatory system of humans and other vertebrates that delivers necessary substances such as nutrients and oxygen to the cells, and transports metabolic waste products away from those same cells.

Venous (darker) and arterial (brighter) blood

Two tubes of EDTA-anticoagulated blood. Left tube: after standing, the RBCs have settled at the bottom of the tube. Right tube: Freshly drawn blood

A scanning electron microscope (SEM) image of a normal red blood cell (left), a platelet (middle), and a white blood cell (right)

Vertebrate red blood cell types, measurements in micrometers