Ductility refers to the ability of a material to sustain significant plastic deformation before fracture. Plastic deformation is the permanent distortion of a material under applied stress, as opposed to elastic deformation, which is reversible upon removing the stress. Ductility is a critical mechanical performance indicator, particularly in applications that require materials to bend, stretch, or deform in other ways without breaking. The extent of ductility can be quantitatively assessed using the percent elongation at break, given by the equation:
Tensile test of an Al-Mg-Si alloy. The local necking and the cup and cone fracture surfaces are typical for ductile metals.
This tensile test of a nodular cast iron demonstrates low ductility.
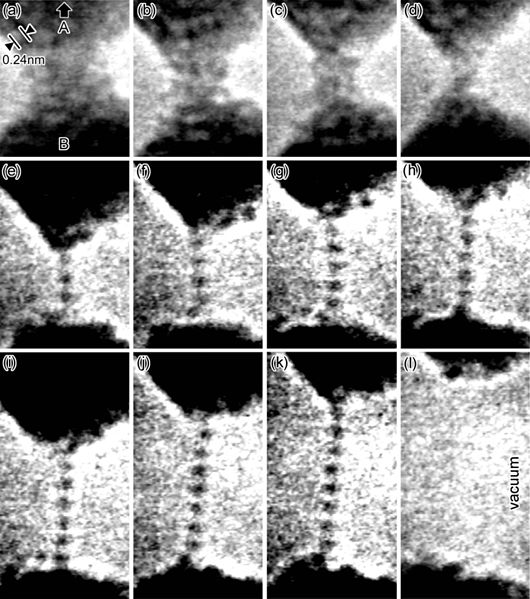
Gold is extremely ductile. It can be drawn into a monatomic wire, and then stretched more before it breaks.
Gold is a chemical element with the symbol Au and the atomic number 79. In its pure form, it is a bright, slightly orange-yellow, dense, soft, malleable, and ductile metal. Chemically, gold is a transition metal, a group 11 element, and one of the noble metals. It is one of the least reactive chemical elements, being the second-lowest in the reactivity series. It is solid under standard conditions.
Gold
Gold can be drawn into a monatomic wire, and then stretched more before it breaks.
A gold nugget of 5 mm (0.20 in) in size can be hammered into a gold foil of about 0.5 m2 (5.4 sq ft) in area.
Gold(III) chloride solution in water