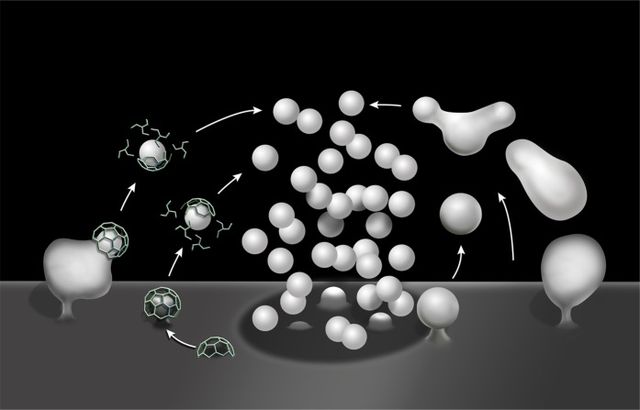
Endocytosis is a cellular process in which substances are brought into the cell. The material to be internalized is surrounded by an area of cell membrane, which then buds off inside the cell to form a vesicle containing the ingested materials. Endocytosis includes pinocytosis and phagocytosis. It is a form of active transport.
Schematic drawing illustrating clathrin-mediated (left) and clathrin-independent endocytosis (right) of synaptic vesicle membranes.
From left to right: Phagocytosis, Pinocytosis, Receptor-mediated endocytosis.
Stage 1
Stage 2
The cell is the basic structural and functional unit of all forms of life. Every cell consists of cytoplasm enclosed within a membrane; many cells contain organelles, each with a specific function. The term comes from the Latin word cellula meaning 'small room'. Most cells are only visible under a microscope. Cells emerged on Earth about 4 billion years ago. All cells are capable of replication, protein synthesis, and motility.
Onion (Allium cepa) root cells in different phases of the cell cycle (drawn by E. B. Wilson, 1900)
A fluorescent image of an endothelial cell. Nuclei are stained blue, mitochondria are stained red, and microfilaments are stained green.
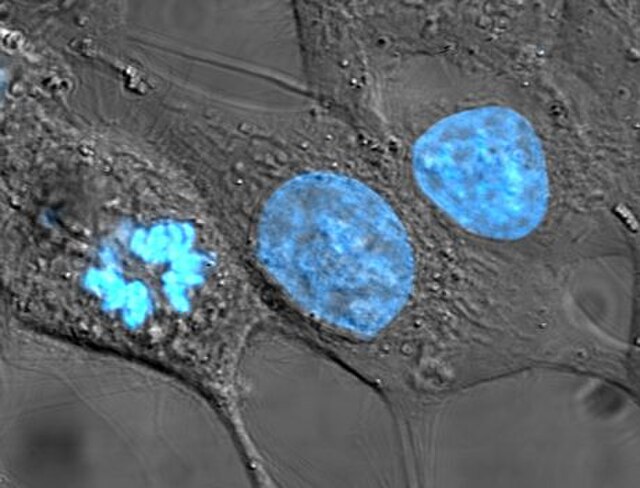
Human cancer cells, specifically HeLa cells, with DNA stained blue. The central and rightmost cell are in interphase, so their DNA is diffuse and the entire nuclei are labelled. The cell on the left is going through mitosis and its chromosomes have condensed.
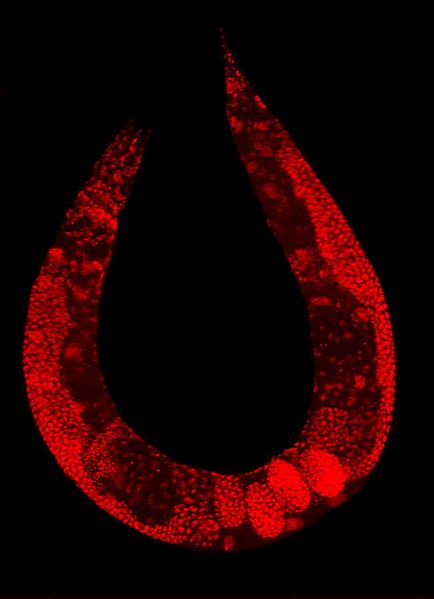
Staining of a Caenorhabditis elegans highlights the nuclei of its cells.