A fuel cell is an electrochemical cell that converts the chemical energy of a fuel and an oxidizing agent into electricity through a pair of redox reactions. Fuel cells are different from most batteries in requiring a continuous source of fuel and oxygen to sustain the chemical reaction, whereas in a battery the chemical energy usually comes from substances that are already present in the battery. Fuel cells can produce electricity continuously for as long as fuel and oxygen are supplied.
Demonstration model of a direct methanol fuel cell (black layered cube) in its enclosure.
Sketch of Sir William Grove's 1839 fuel cell
Condensation of water produced by a PEMFC on the air channel wall. The gold wire around the cell ensures the collection of electric current.
SEM micrograph of a PEMFC MEA cross-section with a non-precious metal catalyst cathode and Pt/C anode. False colors applied for clarity.
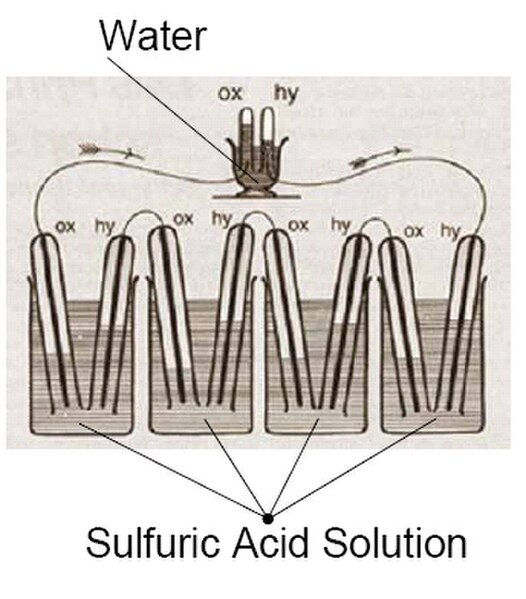
An electrochemical cell is a device that generates electrical energy from chemical reactions. Electrical energy can also be applied to these cells to cause chemical reactions to occur. Electrochemical cells that generate an electric current are called voltaic or galvanic cells and those that generate chemical reactions, via electrolysis for example, are called electrolytic cells.
A modern electrolytic cell consisting of two half reactions, two electrodes, a salt bridge, voltmeter, and a battery.
Circuit diagram of a primary cell showing difference in cell potential, and flow of electrons through a resistor.
Lead acid car battery (secondary cell)