Gasoline direct injection
Videos
Photos
Gasoline direct injection (GDI), also known as petrol direct injection (PDI), is a mixture formation system for internal combustion engines that run on gasoline (petrol), where fuel is injected into the combustion chamber. This is distinct from manifold injection systems, which inject fuel into the intake manifold.

GDI engine from a BMW car (fuel injector is located above the red triangle)

Swirl cavity on the top of a piston in the 2010–2017 Ford EcoBoost 3.5 L engine
Internal combustion engine
Videos
Photos
An internal combustion engine is a heat engine in which the combustion of a fuel occurs with an oxidizer in a combustion chamber that is an integral part of the working fluid flow circuit. In an internal combustion engine, the expansion of the high-temperature and high-pressure gases produced by combustion applies direct force to some component of the engine. The force is typically applied to pistons, turbine blades, a rotor, or a nozzle. This force moves the component over a distance, transforming chemical energy into kinetic energy which is used to propel, move or power whatever the engine is attached to.
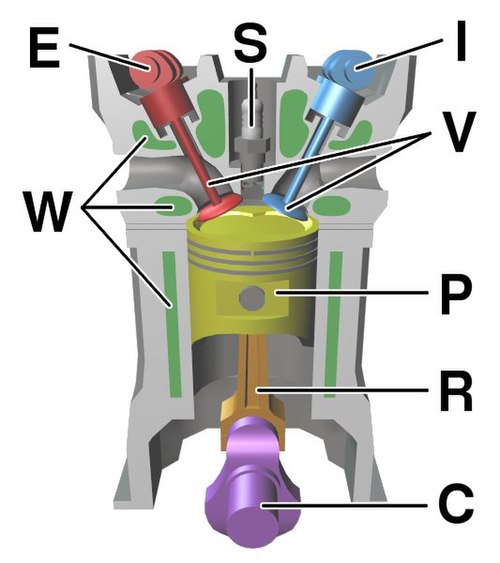
Diagram of a cylinder as found in an overhead cam 4-stroke gasoline engine: C – crankshaft E – exhaust camshaft I – inlet camshaft P – piston R – connecting rod S – spark plug V – valves. red: exhaust, blue: intake. W – cooling water jacket gray structure – engine block

Reciprocating engine of a car

Diesel generator for backup power

Bare cylinder block of a V8 engine