A Geiger counter is an electronic instrument used for detecting and measuring ionizing radiation. It is widely used in applications such as radiation dosimetry, radiological protection, experimental physics and the nuclear industry.
A "two-piece" bench-type Geiger–Müller counter using a cylindrical end-window detector connected to an electronics module with analogue readout
Geiger counter with pancake type probe
Laboratory use of a Geiger counter with end-window probe to measure beta radiation
A Radhound Geiger counter measuring radiation emitted by a tree in Chernobyl
Radiation protection, also known as radiological protection, is defined by the International Atomic Energy Agency (IAEA) as "The protection of people from harmful effects of exposure to ionizing radiation, and the means for achieving this". Exposure can be from a source of radiation external to the human body or due to internal irradiation caused by the ingestion of radioactive contamination.
Large scale glovebox in the nuclear industry used to contain airborne radioactive particles.
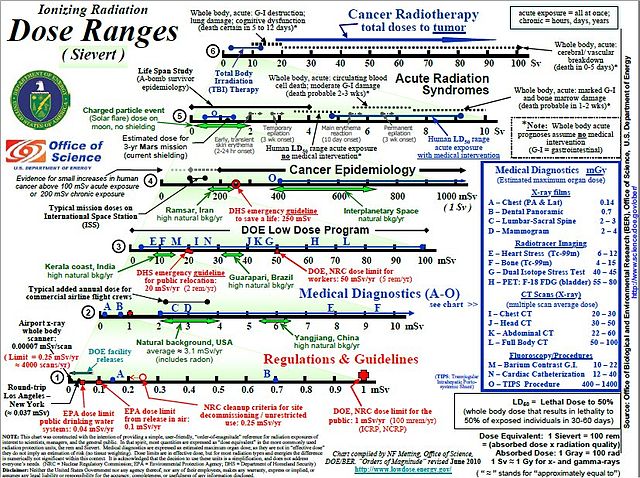
USA Dept of Energy 2010 dose chart in sieverts for a variety of situations and applications.
A lead castle built to shield a radioactive sample in a lab, being a form of lead shielding.
Hand-held ion chamber survey meter in use for surface dose rate on one of three radioisotope thermoelectric generators (RTGs) for the Cassini spacecraft.