An icebox is a compact non-mechanical refrigerator which was a common early-twentieth-century kitchen appliance before the development of safely powered refrigeration devices. Before the development of electric refrigerators, iceboxes were referred to by the public as "refrigerators". Only after the invention of the modern electric refrigerator did early non-electric refrigerators become known as iceboxes. The terms ice box and refrigerator were used interchangeably in advertising as long ago as 1848.
Icebox used in cafés of Paris in the late 1800s
Ice extraction taking place in 1970
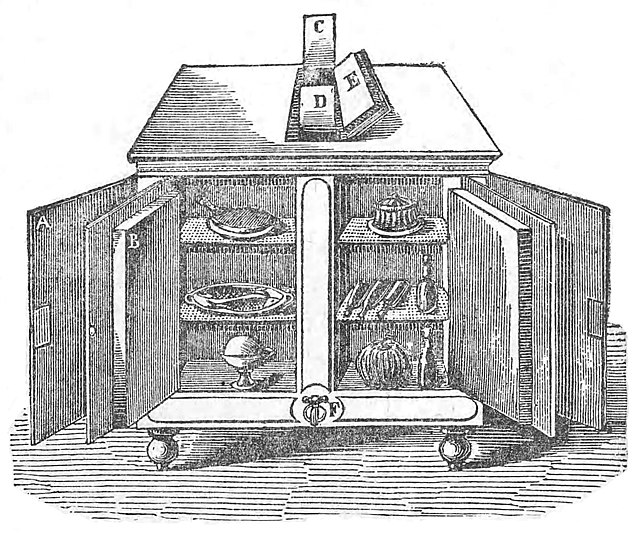
A. Norwegian icebox. The ice was placed in the drawer at top. B. Typical Victorian icebox, of oak with tin or zinc shelving and door lining. C. An oak cabinet icebox that would be found in well-to-do homes.
Women delivering ice. During WWI, many women were employed as icemen (or "ice girls", as they were called).
A refrigerator, colloquially fridge, is a commercial and home appliance consisting of a thermally insulated compartment and a heat pump that transfers heat from its inside to its external environment so that its inside is cooled to a temperature below the room temperature. Refrigeration is an essential food storage technique around the world. The low temperature lowers the reproduction rate of bacteria, so the refrigerator reduces the rate of spoilage. A refrigerator maintains a temperature a few degrees above the freezing point of water. The optimal temperature range for perishable food storage is 3 to 5 °C. A similar device that maintains a temperature below the freezing point of water is called a freezer. The refrigerator replaced the icebox, which had been a common household appliance for almost a century and a half. The United States Food and Drug Administration recommends that the refrigerator be kept at or below 4 °C (40 °F) and that the freezer be regulated at −18 °C (0 °F).

Food in a refrigerator with its door open
Exterior of a modern refrigerator
Ferdinand Carré's ice-making device
Production of refrigerators in France, ca. 1950s