Illite, also called hydromica or hydromuscovite, is a group of closely related non-expanding clay minerals. Illite is a secondary mineral precipitate, and an example of a phyllosilicate, or layered alumino-silicate. Its structure is a 2:1 sandwich of silica tetrahedron (T) – alumina octahedron (O) – silica tetrahedron (T) layers. The space between this T-O-T sequence of layers is occupied by poorly hydrated potassium cations which are responsible for the absence of swelling. Structurally, illite is quite similar to muscovite with slightly more silicon, magnesium, iron, and water and slightly less tetrahedral aluminium and interlayer potassium. The chemical formula is given as (K,H3O)(Al,Mg,Fe)2(Si,Al)4O10[(OH)2·(H2O)], but there is considerable ion (isomorphic) substitution. It occurs as aggregates of small monoclinic grey to white crystals. Due to the small size, positive identification usually requires x-ray diffraction or SEM-EDS analysis. Illite occurs as an altered product of muscovite and feldspar in weathering and hydrothermal environments; it may be a component of sericite. It is common in sediments, soils, and argillaceous sedimentary rocks as well as in some low grade metamorphic rocks. The iron-rich member of the illite group, glauconite, in sediments can be differentiated by x-ray analysis.

Illite
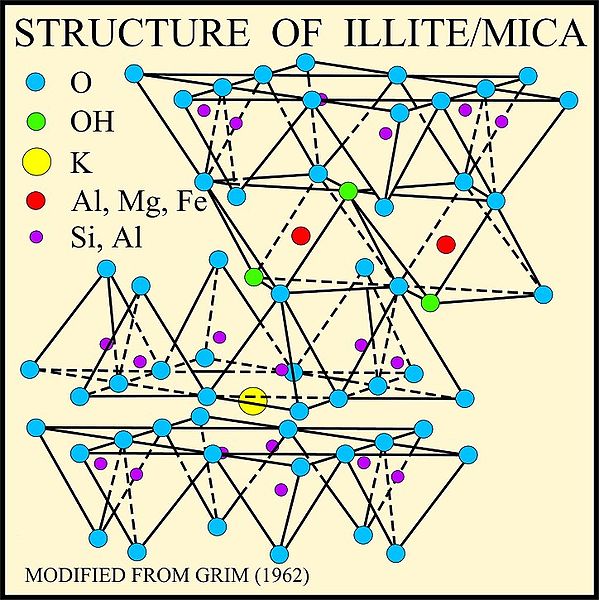
Structure of illite mica – USGS
Clay is a type of fine-grained natural soil material containing clay minerals (hydrous aluminium phyllosilicates, e.g. kaolinite, Al2Si2O5(OH)4). Most pure clay minerals are white or light-coloured, but natural clays show a variety of colours from impurities, such as a reddish or brownish colour from small amounts of iron oxide.
A quaternary clay in Estonia
A 23,500 times magnified electron micrograph of smectite clay
Italian and African-American clay miners in mine shaft, 1910