A mixotroph is an organism that can use a mix of different sources of energy and carbon, instead of having a single trophic mode on the continuum from complete autotrophy at one end to heterotrophy at the other. It is estimated that mixotrophs comprise more than half of all microscopic plankton. There are two types of eukaryotic mixotrophs: those with their own chloroplasts, and those with endosymbionts—and those that acquire them through kleptoplasty or through symbiotic associations with prey or enslavement of their organelles.
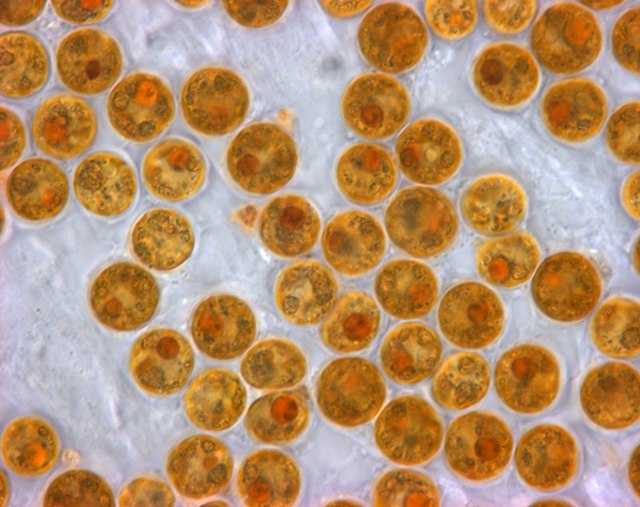
Zooxanthellae is a photosynthetic algae that lives inside hosts like coral
Anthopleura xanthogrammica gains its green colour from Zoochlorella
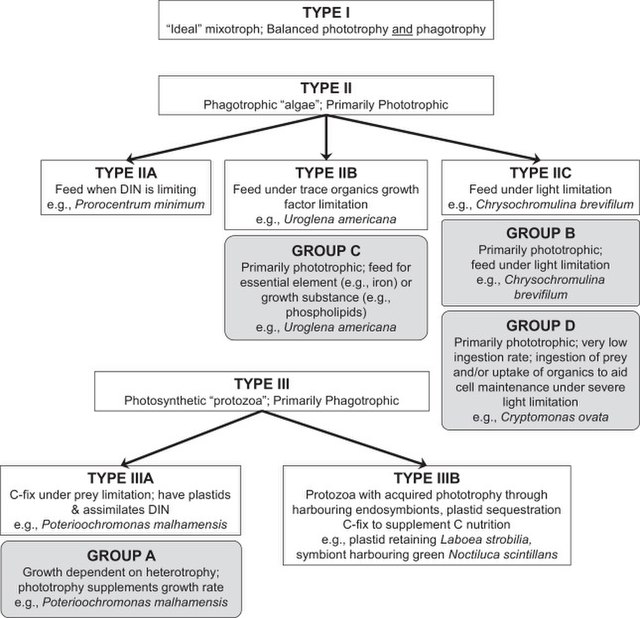
Traditional classification of mixotrophic protists In this diagram, types in open boxes as proposed by Stoecker have been aligned against groups in grey boxes as proposed by Jones. DIN = dissolved inorganic nutrients
Pathways used by Mitra et al. to derive functional groups of planktonic protists
Primary nutritional groups
Primary nutritional groups are groups of organisms, divided in relation to the nutrition mode according to the sources of energy and carbon, needed for living, growth and reproduction. The sources of energy can be light or chemical compounds; the sources of carbon can be of organic or inorganic origin.
Yellow fungus