A monopropellant rocket is a rocket that uses a single chemical as its propellant. Monopropellant rockets are commonly used as small attitude and trajectory control rockets in satellites, rocket upper stages, manned spacecraft, and spaceplanes.
Lunar Landing Research Vehicle with 18 Hydrogen Peroxide Monopropellant Thrusters
Centaur III Upper Stage with 12 Hydrazine Monopropellant Thrusters
A rocket is a vehicle that uses jet propulsion to accelerate without using the surrounding air. A rocket engine produces thrust by reaction to exhaust expelled at high speed. Rocket engines work entirely from propellant carried within the vehicle; therefore a rocket can fly in the vacuum of space. Rockets work more efficiently in a vacuum and incur a loss of thrust due to the opposing pressure of the atmosphere.
A Soyuz-FG rocket launches from "Gagarin's Start" (Site 1/5), Baikonur Cosmodrome
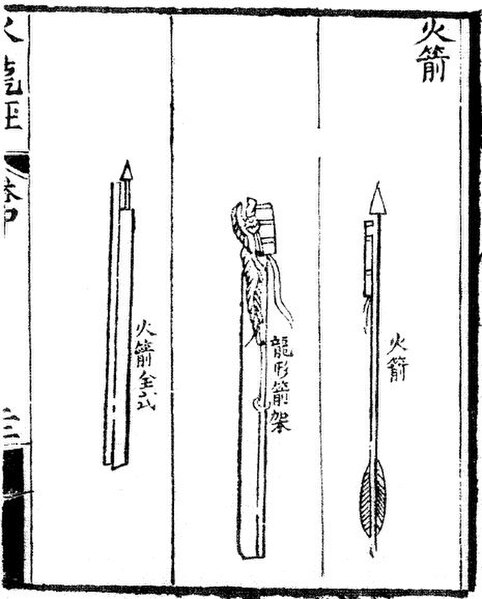
Rocket arrows depicted in the Huolongjing: "fire arrow", "dragon-shaped arrow frame", and a "complete fire arrow"
Mysorean rockets and rocket artillery used to defeat an East India Company battalion during the Battle of Guntur
William Congreve at the bombardment of Copenhagen (1807) during the Napoleonic Wars