Peridotite ( PERR-ih-doh-tyte, pə-RID-ə-) is a dense, coarse-grained igneous rock consisting mostly of the silicate minerals olivine and pyroxene. Peridotite is ultramafic, as the rock contains less than 45% silica. It is high in magnesium (Mg2+), reflecting the high proportions of magnesium-rich olivine, with appreciable iron. Peridotite is derived from Earth's mantle, either as solid blocks and fragments, or as crystals accumulated from magmas that formed in the mantle. The compositions of peridotites from these layered igneous complexes vary widely, reflecting the relative proportions of pyroxenes, chromite, plagioclase, and amphibole.
Typical peridotite sample (dunite, left) and large olivine crystal (right)
Olivine in a peridotite weathering to iddingsite within a mantle xenolith
Serpentinized and carbonated peridotite
Alpine peridotite from the Ivrea zone in the Alps of Italy (dunite from Finero)
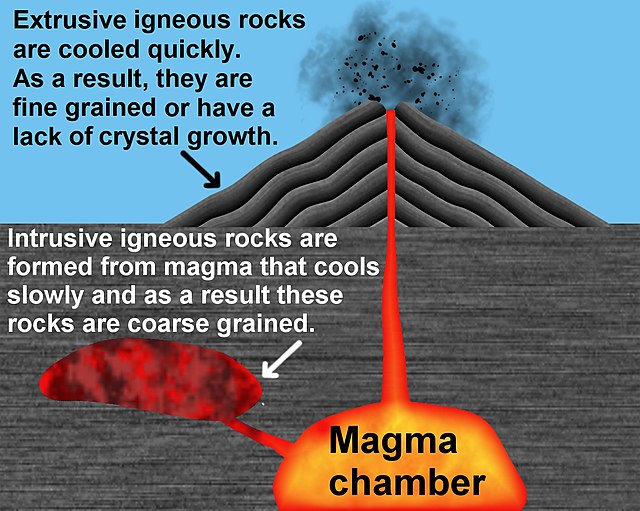
Igneous rock, or magmatic rock, is one of the three main rock types, the others being sedimentary and metamorphic. Igneous rocks are formed through the cooling and solidification of magma or lava.
Geologic provinces of the world (USGS) Shield Platform Orogen Basin Large igneous province Extended crust Oceanic crust: 0–20 Ma 20–65 Ma >65 Ma
Volcanic eruptions of lava are major sources of igneous rocks. (Mayon volcano in the Philippines, erupting in 2009)
Natural columns of igneous rock separated from each other by columnar joints, in Madeira
Formation of igneous rock