Silver iodide
Videos
Page
Silver iodide is an inorganic compound with the formula AgI. The compound is a bright yellow solid, but samples almost always contain impurities of metallic silver that give a grey colouration. The silver contamination arises because some samples of AgI can be highly photosensitive. This property is exploited in silver-based photography. Silver iodide is also used as an antiseptic and in cloud seeding.

Silver iodide

The golden-yellow crystals on this mineral sample are iodargyrite, a naturally occurring form of β-AgI.

Cessna 210 equipped with a silver iodide generator for cloud seeding
Silver
Videos
Page
Silver is a chemical element; it has symbol Ag ) and atomic number 47. A soft, white, lustrous transition metal, it exhibits the highest electrical conductivity, thermal conductivity, and reflectivity of any metal. The metal is found in the Earth's crust in the pure, free elemental form, as an alloy with gold and other metals, and in minerals such as argentite and chlorargyrite. Most silver is produced as a byproduct of copper, gold, lead, and zinc refining.

Silver
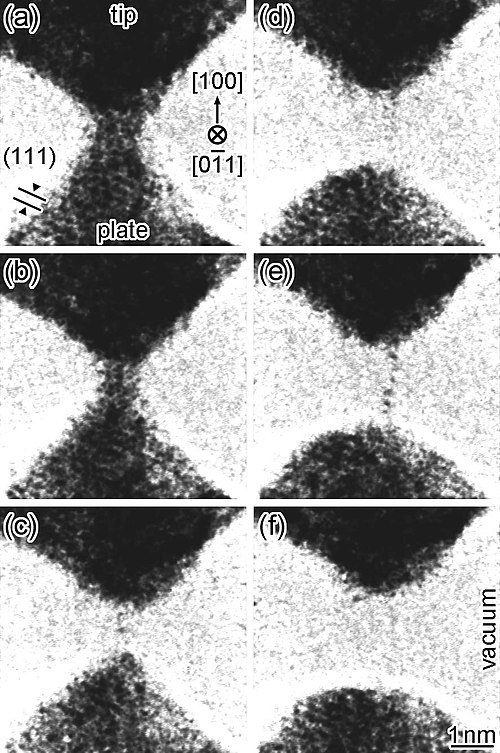
Silver is extremely ductile, and can be drawn into a wire one atom wide.

The three common silver halide precipitates: from left to right, silver iodide, silver bromide, and silver chloride

Crystals of silver nitrate