Visual phototransduction is the sensory transduction process of the visual system by which light is detected by photoreceptor cells in the vertebrate retina. A photon is absorbed by a retinal chromophore, which initiates a signal cascade through several intermediate cells, then through the retinal ganglion cells (RGCs) comprising the optic nerve.
The absorption of light leads to an isomeric change in the retinal molecule.
The visual system is the physiological basis of visual perception. The system detects, transduces and interprets information concerning light within the visible range to construct an image and build a mental model of the surrounding environment. The visual system is associated with the eye and functionally divided into the optical system and the neural system.
The eye is the sensory organ of the visual system. The iris, pupil, and sclera are visible
Representation of optic pathways from each of the 4 quadrants of view for both eyes simultaneously
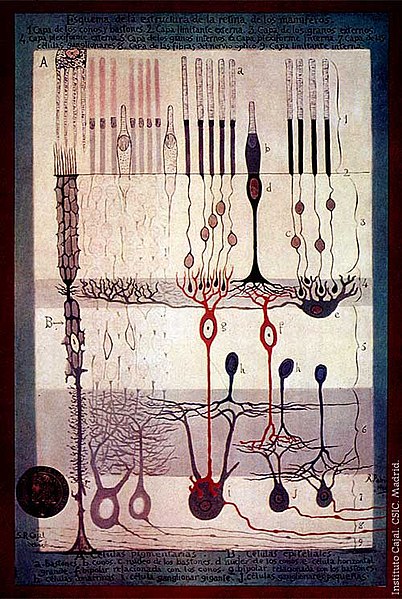
S. Ramón y Cajal, Structure of the Mammalian Retina, 1900
Information flow from the eyes (top), crossing at the optic chiasma, joining left and right eye information in the optic tract, and layering left and right visual stimuli in the lateral geniculate nucleus. V1 in red at bottom of image. (1543 image from Andreas Vesalius' Fabrica)