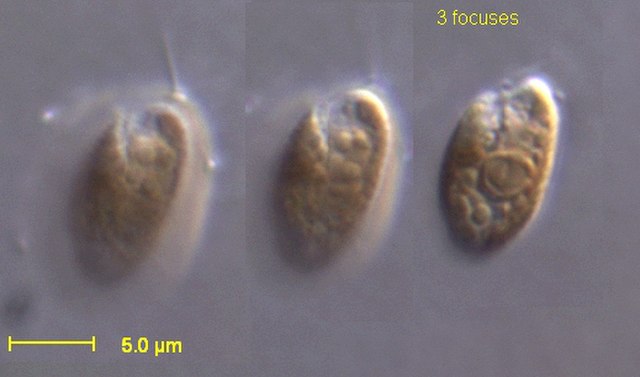
The cryptomonads are a group of algae, most of which have plastids. They are common in freshwater, and also occur in marine and brackish habitats. Each cell is around 10–50 μm in size and flattened in shape, with an anterior groove or pocket. At the edge of the pocket there are typically two slightly unequal flagella.
Cryptomonad
Cryptophytes under SEM
Cryptophytes under light microscope
Algae are any of a large and diverse group of photosynthetic, eukaryotic organisms. The name is an informal term for a polyphyletic grouping that includes species from multiple distinct clades. Included organisms range from unicellular microalgae, such as Chlorella, Prototheca and the diatoms, to multicellular forms, such as the giant kelp, a large brown alga which may grow up to 50 metres (160 ft) in length. Most are aquatic and lack many of the distinct cell and tissue types, such as stomata, xylem and phloem that are found in land plants. The largest and most complex marine algae are called seaweeds, while the most complex freshwater forms are the Charophyta, a division of green algae which includes, for example, Spirogyra and stoneworts. Algae that are carried by water are plankton, specifically phytoplankton.
Image: NSW seabed 1
Image: Водоросли пресноводного водоема 2
False-color scanning electron micrograph of the unicellular coccolithophore Gephyrocapsa oceanica
The kelp forest exhibit at the Monterey Bay Aquarium: A three-dimensional, multicellular thallus