In vacuum tubes and gas-filled tubes, a hot cathode or thermionic cathode is a cathode electrode which is heated to make it emit electrons due to thermionic emission. This is in contrast to a cold cathode, which does not have a heating element. The heating element is usually an electrical filament heated by a separate electric current passing through it. Hot cathodes typically achieve much higher power density than cold cathodes, emitting significantly more electrons from the same surface area. Cold cathodes rely on field electron emission or secondary electron emission from positive ion bombardment, and do not require heating. There are two types of hot cathode. In a directly heated cathode, the filament is the cathode and emits the electrons. In an indirectly heated cathode, the filament or heater heats a separate metal cathode electrode which emits the electrons.

A tungsten filament acting as a directly heated cathode in a low pressure mercury gas discharge lamp which emits electrons. To increase electron emission, a white thermionic emission mix coating is applied on hot cathodes, visible on the central portion of the coil. Typically made of a mixture of barium, strontium, and calcium oxides, the coating is sputtered away through normal use, eventually resulting in lamp failure.
Two indirectly heated cathodes (orange heater strip) in ECC83 dual triode tube
Glow of a directly heated cathode in an Eimac 4-1000A 1 kW power tetrode tube in a radio transmitter. Directly heated cathodes operate at higher temperatures and produce a brighter glow. The cathode is behind the other tube elements and not directly visible.
SEM Image of G1 Support and G1 Wire, of a heavily used Pentode showing Barium oxide Contamination (green) from the cathode.
A vacuum tube, electron tube, valve, or tube, is a device that controls electric current flow in a high vacuum between electrodes to which an electric potential difference has been applied.
Later thermionic vacuum tubes, mostly miniature style, some with top cap connections for higher voltages
Hot tubes in an audio power amplifier, emitting their distinctive red-orange glow
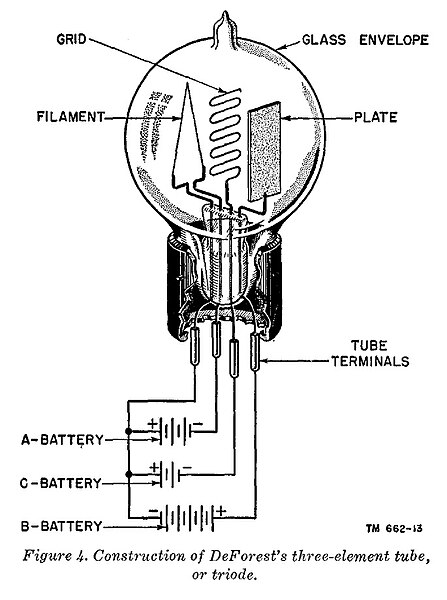
Illustration representing a primitive triode vacuum tube and the polarities of the typical DC operating potentials. Not shown are the impedances (resistors or inductors) that would be included in series with the C and B voltage sources.
Radio station signal generator with vacuum tubes