Lunar Orbiter program
Videos
Page
The Lunar Orbiter program was a series of five uncrewed lunar orbiter missions launched by the United States from 1966 through 1967. Intended to help select Apollo landing sites by mapping the Moon's surface, they provided the first photographs from lunar orbit and photographed both the Moon and Earth.

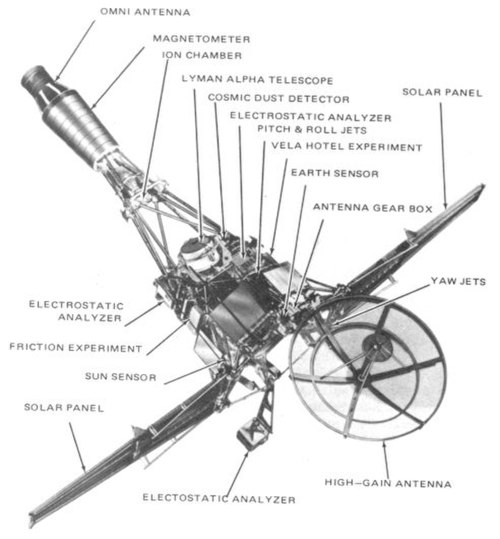
Lunar Orbiter spacecraft

Lunar Orbiter camera (NASA)

A detail of an original image at the top, compared to a reprocessed version at the bottom created by LOIRP.

Image: NASM A19700318000 NASM2019 06087
Atlas-Agena
Videos
Page
The Atlas-Agena was an American expendable launch system derived from the SM-65 Atlas missile. It was a member of the Atlas family of rockets, and was launched 109 times between 1960 and 1978. It was used to launch the first five Mariner uncrewed probes to the planets Venus and Mars, and the Ranger and Lunar Orbiter uncrewed probes to the Moon. The upper stage was also used as an uncrewed orbital target vehicle for the Gemini crewed spacecraft to practice rendezvous and docking. However, the launch vehicle family was originally developed for the Air Force and most of its launches were classified DoD payloads.

An Atlas-Agena launching Lunar Orbiter 4
Ranger block I spacecraft bus was used for the first two Rangers, and also for the first two Mariner interplanetary probes

Mariner 3, 4, and 5 spacecraft bus

The Agena Target Vehicle as seen from Gemini 8 during rendezvous, March 16, 1966