Pressure
Videos
Photos
Pressure is the force applied perpendicular to the surface of an object per unit area over which that force is distributed. Gauge pressure is the pressure relative to the ambient pressure.

Mercury column

The effects of an external pressure of 700 bar on an aluminum cylinder with 5 mm (0.197 in) wall thickness

Water escapes at high speed from a damaged hydrant that contains water at high pressure

Low-pressure chamber in Bundesleistungszentrum Kienbaum, Germany
Force
Videos
Photos
In physics, a force is an influence that can cause an object to change its velocity, i.e., to accelerate, meaning a change in speed or direction, unless counterbalanced by other forces. The concept of force makes the everyday notion of pushing or pulling mathematically precise. Because the magnitude and direction of a force are both important, force is a vector quantity. The SI unit of force is the newton (N), and force is often represented by the symbol F.

Aristotle famously described a force as anything that causes an object to undergo "unnatural motion"

Sir Isaac Newton in 1689. His Principia presented his three laws of motion in geometrical language, whereas modern physics uses differential calculus and vectors.

Galileo Galilei was the first to point out the inherent contradictions contained in Aristotle's description of forces.
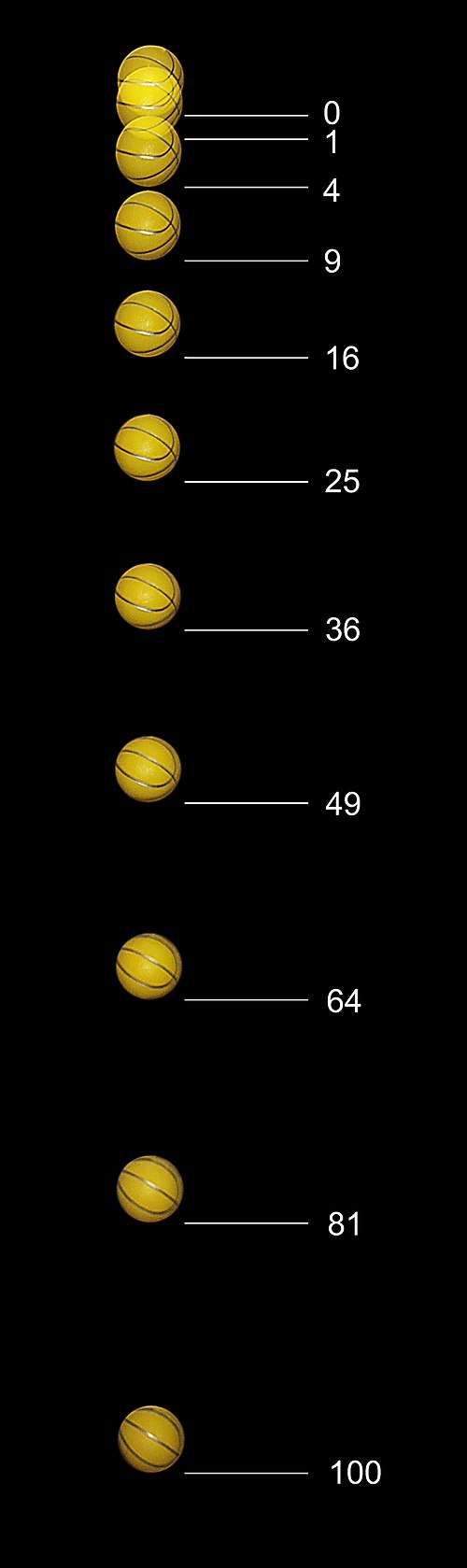
Images of a freely falling basketball taken with a stroboscope at 20 flashes per second. The distance units on the right are multiples of about 12 millimeters. The basketball starts at rest. At the time of the first flash (distance zero) it is released, after which the number of units fallen is equal to the square of the number of flashes.