The Stark effect is the shifting and splitting of spectral lines of atoms and molecules due to the presence of an external electric field. It is the electric-field analogue of the Zeeman effect, where a spectral line is split into several components due to the presence of the magnetic field. Although initially coined for the static case, it is also used in the wider context to describe the effect of time-dependent electric fields. In particular, the Stark effect is responsible for the pressure broadening of spectral lines by charged particles in plasmas. For most spectral lines, the Stark effect is either linear or quadratic with a high accuracy.
Computed energy level spectrum of hydrogen as a function of the electric field near n = 15 for magnetic quantum number m = 0. Each n level consists of n − 1 degenerate sublevels; application of an electric field breaks the degeneracy. Energy levels can cross due to underlying symmetries of motion in the Coulomb potential.
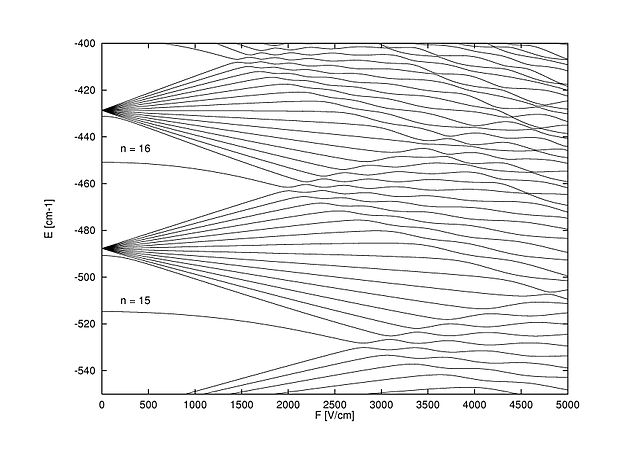
Lithium Rydberg-level spectrum as a function of the electric field near n = 15 for m = 0. Note how a complicated pattern of the energy levels emerges as the electric field increases, not unlike bifurcations of closed orbits in classical dynamical systems leading to chaos.
A spectral line is a weaker or stronger region in an otherwise uniform and continuous spectrum. It may result from emission or absorption of light in a narrow frequency range, compared with the nearby frequencies. Spectral lines are often used to identify atoms and molecules. These "fingerprints" can be compared to the previously collected ones of atoms and molecules, and are thus used to identify the atomic and molecular components of stars and planets, which would otherwise be impossible.
Continuous spectrum of an incandescent lamp (mid) and discrete spectrum lines of a fluorescent lamp (bottom)