Thermodynamic temperature
Thermodynamic temperature is a quantity defined in thermodynamics as distinct from kinetic theory or statistical mechanics.
Figure 6 Ice and water: two phases of the same substance
Figure 8 When many of the chemical elements, such as the noble gases and platinum-group metals, freeze to a solid — the most ordered state of matter — their crystal structures have a close-packed arrangement. This yields the greatest possible packing density and the lowest energy state.
Figure 9 Due to the effects of zero-point energy, helium at ambient pressure remains a superfluid even when exceedingly close to absolute zero; it won't freeze unless under 25 bar of pressure (~25 atmospheres).
Anders Celsius
Thermodynamics is a branch of physics that deals with heat, work, and temperature, and their relation to energy, entropy, and the physical properties of matter and radiation. The behavior of these quantities is governed by the four laws of thermodynamics, which convey a quantitative description using measurable macroscopic physical quantities, but may be explained in terms of microscopic constituents by statistical mechanics. Thermodynamics applies to a wide variety of topics in science and engineering, especially physical chemistry, biochemistry, chemical engineering and mechanical engineering, but also in other complex fields such as meteorology.
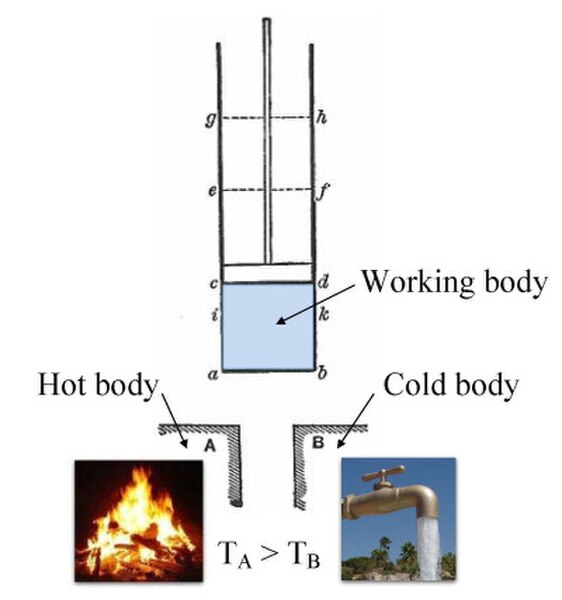
Annotated color version of the original 1824 Carnot heat engine showing the hot body (boiler), working body (system, steam), and cold body (water), the letters labeled according to the stopping points in Carnot cycle
Opening a bottle of sparkling wine (high-speed photography). The sudden drop of pressure causes a huge drop of temperature. The moisture in the air freezes, creating a smoke of tiny ice crystals.