X-ray crystallography is the experimental science of determining the atomic and molecular structure of a crystal, in which the crystalline structure causes a beam of incident X-rays to diffract in specific directions. By measuring the angles and intensities of the X-ray diffraction, a crystallographer can produce a three-dimensional picture of the density of electrons within the crystal and the positions of the atoms, as well as their chemical bonds, crystallographic disorder, and other information.
A powder X-ray diffractometer in motion
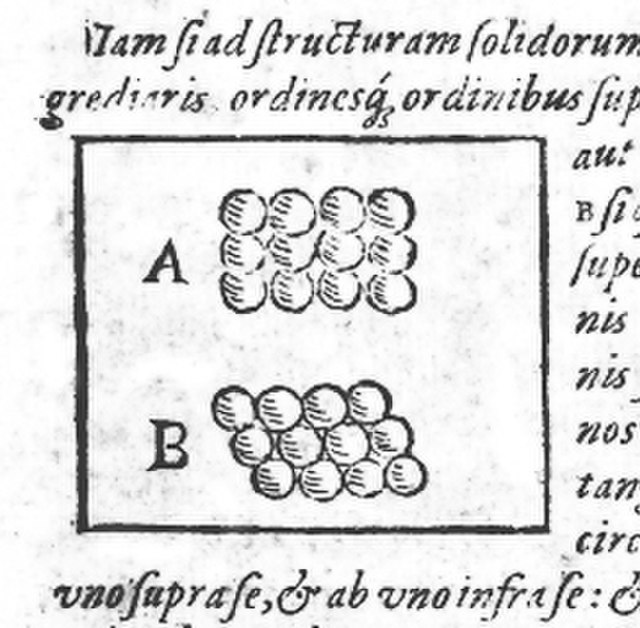
Drawing of square (A) and hexagonal (B) packing from Kepler's work, Strena seu de Nive Sexangula.
One of the copper sulfate X-ray interference patterns published in Von Laue's 1912 paper.
Although diamonds (top left) and graphite (top right) are identical in chemical composition—being both pure carbon—X-ray crystallography revealed the arrangement of their atoms (bottom). In diamond, the carbon atoms are arranged tetrahedrally and held together by single covalent bonds. By contrast, graphite is composed of stacked sheets. Within the sheet, the bonding is covalent and has hexagonal symmetry, but there are no covalent bonds between the sheets.
Diffraction is the interference or bending of waves around the corners of an obstacle or through an aperture into the region of geometrical shadow of the obstacle/aperture. The diffracting object or aperture effectively becomes a secondary source of the propagating wave. Italian scientist Francesco Maria Grimaldi coined the word diffraction and was the first to record accurate observations of the phenomenon in 1660.
A diffraction pattern of a red laser beam projected onto a plate after passing through a small circular aperture in another plate
Single-slit diffraction in a circular ripple tank
Computer-generated intensity pattern formed on a screen by diffraction from a square aperture
Optical diffraction pattern (laser, analogous to X-ray diffraction)